CRISPR/Cas-9
What it is:
Naturally occurring adaptive immunity in bacteria
The CRISPR/Cas-9 system is how bacterial immune systems learn from past experience. Bacteria use CRISPR to remember viral invaders so that Cas9 can attack them the next time they pose a threat. Bacteria can do this by specifically recognizing the invader’s DNA and cutting it up.
CRISPR stands for clustered regularly interspaced short palindromic repeats, areas where a DNA sequence is repeated and separated by “spacer DNA”. The CRISPR region functions like a working memory for bacteria, where the sequences of new invaders are added so a bacterium can recognize them as invading genetic material and eliminate them if ever encountered again. Once foreign genetic sequences are inserted in the CRISPR region, they are transcribed into small CRISPR RNAs (crRNA). crRNAs will bind to any invader that carries matching sequences, flagging foreign DNA to be destroyed. Cas9 is the enzyme that carries out the invader destruction: it is an endonuclease (an enzyme that cuts in the middle of a DNA strand) that cuts DNA wherever the crRNA guides it. In short, CRISPR is a region of the bacterial genome that can keep track of past invaders. Its crRNA product recognizes those invaders whenever they enter the cell again. Cas9 is the enzyme that cuts the foreign DNA, guided by crRNAs.
How it is used:
Editing the genome
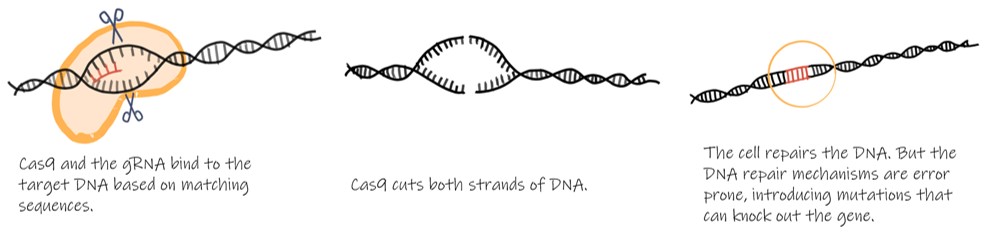
Scientists often spend a lot of time and energy trying to turn off specific genes so that we can better understand their functions. This can be difficult and involves a lot of trial and a lot of error. But recently, scientists have found a way to use the CRISPR/Cas9 system to make this task easier and more precise. The technique takes advantage of Cas9’s ability to work as DNA scissors that we can target to any gene as long as we can make RNA to guide it. Researchers can target Cas9 to cut a specific gene by inserting “guide RNA” (gRNA) with the target gene’s sequence. Cas9 will only cut where the gRNA binds. Researchers can now specifically “cut” any given gene. The gene won’t stay cut forever, though. The cell’s repair mechanisms will rejoin cut ends of the DNA, but these repair mechanisms tend to make mistakes, introducing mutations. The introduced mutations will often knock out the function of the gene entirely. Scientists can then look at what happens to the organism when the gene is no longer functional and learn about its role in the cell.
Scientists have also developed ways to use CRISPR/Cas9 to make specific and very precise changes to a target gene. CRISPR/Cas9 on its own is designed to destroy DNA, but cells have a second more precise DNA repair mechanism that can mend the cut according to a DNA template. This process is called homology directed repair. Homology directed repair relies on the cell fixing damaged DNA using a repair template, a small section of DNA that has portions of sequence which match the ends of the cut strands. If scientist can provide the repair template that the cell uses, then they can change a target gene to their specifications. That’s exactly what scientists have learned to do using CRISPR/Cas9. Along with Cas9 and the gRNA to target the cut, scientists add a DNA repair template which has the sequence of the target gene, but with desired edits in the middle. The repair template becomes part of the host’s DNA when the cell repairs the cut with the changes the scientist wants included. This way we can use CRISPR/Cas9 to very precisely change the sequence of genes instead of just turning them off!
The future:
Editing out disease and debates about editing the germline
CRISPR/Cas9 technology makes it much easier to modify traits in experimental animals as scientists now have the power to make specific genetic changes in genomic DNA with more precision than ever before. CRISPR/Cas9 has also allowed scientists to apply that potential to a much greater number of species beyond the mice, flies, and worms traditionally used for genetic manipulation in the lab. The potential uses of this technology are wide ranging and the speed at which scientists are taking advantage of those uses has many calling CRISPR/Cas9 a revolutionary turning point in the field of genetics. From creating mosquitoes that cannot spread malaria, to pigs whose organs may be more suitable for human transplantation, the CRISPR/Cas9 revolution seems to be just getting going.
More controversially, CRISPR/Cas9 also has the potential to “edit out” genetic mutations that can trigger disease in humans. Diseases like cystic fibrosis, sickle cell anemia, and many others are debilitating and deadly diseases that are caused by inherited mutations. CRISPR/Cas9 has the potential to treat such diseases by directly fixing the mutations causing them. It also has the potential to let parents change the DNA of an embryo long before mutations cause disease in their baby. By fixing the mutation in an embryo, these genetic diseases can be permanently removed from their family tree. The ability to edit human DNA has opened arguments about what ethical guidelines must accompany such research. In 2018, a Chinese researcher announced that the first babies had been born whose genomes had been modified using CRISPR/Cas9. The scientist had not followed the typical steps in gaining approval for scientific trials on humans, and the announcement was met with widespread condemnation from the scientific community. Adding to the controversy, the scientist had not “fixed” a disease mutation, but had instead disabled a gene that makes people susceptible to HIV. By doing this he had tried to improve on otherwise genetically normal individuals. While in this case, most people agree the technology was applied inappropriately, many people think making certain types of genetic modifications in humans is acceptable. Others argue any use editing human DNA is taking the power of CRISPR/Cas9 a step too far.
Learn more:
- “CRISPR: Gene editing and beyond” Nature Videos.
- “CRISPR/Cas9 and Targeted Genome Editing: A New Era in Molecular Biology.” New England Biolabs Inc.
Questions
Review:
-
Why have bacteria evolved enzymes that recognize and cut DNA? Does this relate to any general functions of human body systems?
-
CRISPR/Cas9 is composed of two basic components, CRISPR and Cas9. Describe the function of each separately.
-
Disease is a very broad term that encompasses many types of illnesses. What types of diseases could CRISPR/Cas9 potentially be used to treat? For what types of diseases would CRISPR/Cas9 not be effective?
-
Before CRISPR/Cas9, editing an organism’s genome often involved inserting DNA almost randomly into the organism’s genome. In this way, the scientist could control what was being inserted, but not where. How does this specifically differ from CRISPR/Cas9? What risks might it entail?
-
Why is altering somatic cells, or non-reproductive cells, considered different from altering reproductive cells? Why would editing reproductive cells be considered more controversial?
Critical thinking:
-
Based on the information you have read, would you consider CRISPR/Cas9 to be a discovery by scientists or an invention of new technology?
-
Do you think CRISPR/Cas9 could be used to alter chromosomal rearrangements like are found in individuals with Down’s syndrome, Turner syndrome or Fragile X syndrome? Why or why not?
Discussion:
-
Propose your own guidelines for how CRISPR/Cas9 should be allowed to be used. Should people be allowed to alter any gene they want; after all, it is their own DNA? Should use of CRISPR/Cas9 be highly restricted to use on known, severe diseases? Should it only be allowed to be used on somatic cells and not reproductive cells? Should we not be altering human DNA at all?
Answer key:
Available to teachers upon request: dnadots@minipcr.com
